Réglementation turque sur les produits chimiques
Türkiye REACH (KKDIK)
Qu'est-ce que KKDIK (Türkiye REACH) ?
KKDIK came into force à la suite de the EU Adaptation policy. The first phase of pre-registration ended on December 31st, 2020. The registration phase then officially started the next day (January 1st, 2021). By complying with KKDIK, exporters can ensure their access to the Turkish Market. Exporters who wish to enter the Turkish market but missed the pre-registration should pre-register as soon as possible to be able to benefit from the advantages of joint registration.
KKDIK (aka Türkiye REACH) came into force on the 23rd of June 2017 and has replaced three existing regulations on chemicals and mixtures to regulate the chemical inventory, enforce secure usage, and risk management on hazardous substances, and promote alternatives to hazardous substances or procedures.
The regulation provides the means of registration, evaluation, authorization, and restrictions of chemicals and safeguarding human health and the environment. It also encourages using alternative methods for evaluating the hazard of substances such as decreasing the number of animal tests and encouraging innovation so that the competitiveness of the Turkish chemical industry will be enhanced.
Délais actuels
The Ministry of Environment, Urbanization, and Climate Change of Türkiye announced new KKDIK deadlines for the registration process.
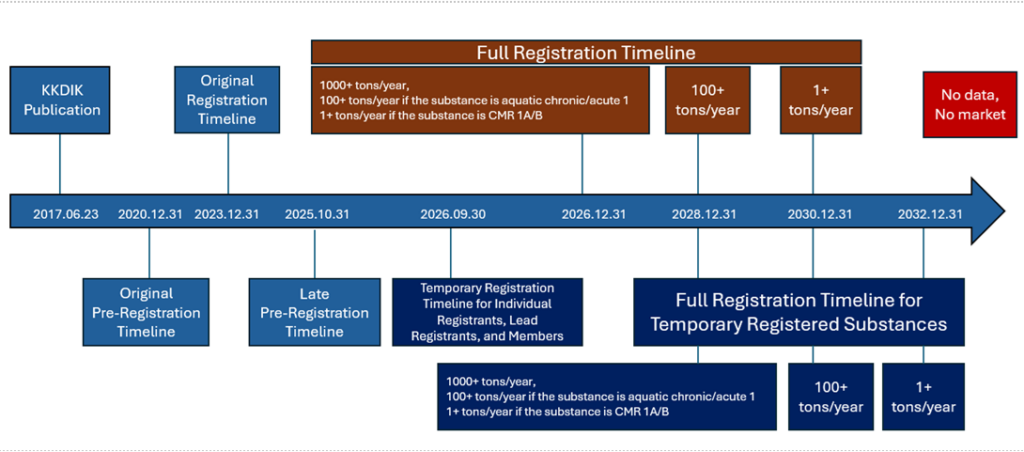
Le calendrier de KKDIK se compose de trois phases principales :
Pre-registration (Deadline has passed):
The deadline set by the Ministry for pre-registration has passed. However, the system continues to accept pre-registrations, and pre-registration must be completed as soon as possible.
Temporary Registration (30 September 2026):
If there are companies that will not be able to complete the full registration process on time, they may submit a temporary registration dossier. Regardless of whether they are individual registrants, lead registrants, or members, and irrespective of tonnage, all temporary registration dossiers must be submitted to the Ministry by 30 September 2026. Companies submitting a temporary registration dossier are not exempt from full registration; they are still required to submit the full registration dossier at a later stage.
Note 1: If the necessary data for full registration cannot be obtained for substances that have undergone temporary registration, an additional two-year extension will be granted to the registration deadlines.
Note 2: Companies that have already submitted a full registration dossier cannot submit a temporary registration dossier.
Note 3: Companies may also choose to submit only the full registration dossier without submitting a temporary registration dossier in advance. Temporary registration is not mandatory.
Full Registration:
a) Until 31/12/2026, for substances meeting the following conditions:
- Substances manufactured or imported in quantities of 1,000 tonnes or more per year, on their own, in mixtures, or in articles,
- Substances manufactured or imported in quantities of 100 tonnes or more per year, on their own, in mixtures, or in articles, and classified as Aquatic Acute 1 and/or Aquatic Chronic 1 (H400, H410) according to the Regulation on the Classification, Labelling and Packaging of Substances and Mixtures,
- Substances manufactured or imported in quantities of 1 tonne or more per year, on their own, in mixtures, or in articles, and classified as carcinogenic, mutagenic, and/or toxic to reproduction Category 1A or 1B according to the Regulation on the Classification, Labelling and Packaging of Substances and Mixtures.
b) Without prejudice to the period specified in point (a) of this paragraph, until31/12/2028 for substances manufactured or imported in quantities of 100 tonnes or more per year, on their own, in mixtures, or in articles,
c) Without prejudice to the periods specified in points (a) and (b) of this paragraph, until31/12/2030 for substances manufactured or imported in quantities of 1 tonne or more per year, on their own, in mixtures, or in articles, shall apply.
La phase d'inscription à Türkiye Reach (KKDIK) a commencé !
Türkiye REACH (KKDIK) Registration Services By GPC Regulatory
KKDIK registration through GPC Regulatory transforms regulatory complexity into a streamlined market gateway. As your trusted KKDIK registration consultant, we deliver seamless customs clearance, preferred supplier status with Turkish buyers, reduced costs via joint registration, and uninterrupted market access through 2030. We handle the regulation—you focus on your business. One partner, complete compliance, lasting market security.
Our KKDIK Compliance Services
1. Only Representative (OR) Services
For non-Turkish manufacturers
- There are no substances classified as hazardous that can be registered under the KKDIK in Turkey. Companies based in the EU Member States also need an Only Representative (OR) to register substances.
- Legal representation in Turkey (KKS system)
- Direct liaison with Turkish authorities (MoEUCC)
- Management of downstream user obligations
- Full confidentiality of sensitive data
2. Late Pre-registration & SIEF Management
- The 2020 Census deadline has now passed on 30 September 2020 but it is still important to register with UK authorities.
Our Support Includes
- 48-hour submission of Late Pre-registration
- Full management of KKS system filings
- Representation in SIEFs (MBDF)
- Negotiation of fair data-sharing costs
3. Lead Registrant (LR) Services
- For high volume or high risk chemicals, ChemNet strongly recommends that a Lead Registrant be selected.
Our Advantage
- 400+ Lead Registrations completed
- Access to extensive existing data
- Faster and more cost-efficient registration
We Handle
- Joint dossier preparation (CSR included)
- SIEF coordination
- Cost-sharing agreements
- Temporary registration submissions
4. Turkish Safety Data Sheet (SDS / GBF)
- Regulatory Requirements
- Must be in Turkish language
- Certified by a KDU expert
- Uploaded to official MoEUCC system
Our Solution
- Fully compliant SDS preparation
- Translation + certification
- Continuous updates as regulations evolve
5. Authorization & Restriction Compliance
- For SVHCs and restricted substances:
- Authorization applications (Annex XIV)
- Restriction audits (Annex XVII)
Supply chain communication compliance.
Why Choose GPC Regulatory as Your Turkey REACH Registration Consultant?
The choice of compliance provider is fundamental to cost, speed and market access. As a leading Turkey REACH registration consultant, GPC Regulatory supports companies to meet their regulatory obligations on time and cost effectively.
1. Proven Track Record
As a preeminent firm in the field of EU REACH, GPC Regulatory has managed well over 1,200 registration dossiers and undertaken over 400 Lead Registrant roles. We now bring this unique expertise to the KKDIK regulations in Turkey.
1,200+ registration dossiers successfully managed
400+ Lead Registrant roles under EU REACH
48-hour turnaround for Late Pre-registration
2. Speed You Can Rely On
The various regulatory submission dates are rapidly approaching, and we can deliver a 48-hour pre-registration filing or accelerated dossier submission in a variety of languages.
48-hour pre-registration turnaround
Accelerated dossier preparation
Avoid delays and missed deadlines
We can act as Représentant unique (RO) in Turkey and handle the pre-registration and full dossier submission as well as SDS.
3. True Turkey REACH Registration Support
Unlike other competitors in the market, GPC Regulatory offers a one stop solution for KKDIK compliance for Turkey. Unlike others who only deal with one part of the solution, GPC can manage the whole process for your company from pre-registration in 2018 all the way to full registration in 2030.
Based on the progress in pre-registration, the SIEF negotiation, temporary registration and planned full registration in 2030.
- One partner. Complete compliance.
- No handoffs, no gaps, no surprises
4. Local Presence in Turkey
Our main office of GPC Regulatory is located in Turkey where our team is easily accessible to the MoEUCC and the KKS system in process plant. You will get immediate response to your questions and be able to resolve your issues quickly. We cannot find any offshore competitor who can do the same.
- Direct access to MoEUCC and KKS system
- Faster issue resolution
- No communication delays or time zone barriers
- Certified Regulatory Experts
Our team consists of KDU certified SDS authors and Chemical Safety Assessors who are qualified to sign off dossiers in Turkey. Our team has a strong understanding of the legislative requirements but also real scientific expertise.
- KDU-certified SDS authors on staff
- Deep regulatory + scientific expertise
- Fully compliant, audit-ready documentation
5. Strategic Turkey REACH Registration Consulting Services
Unlike many other compliance providers, who simply submit various documents to relevant authorities, GPC Regulatory also can optimize a company’s long-term cost, strategy, and even competitiveness by preparing it for submission of REACH registrations to the Turkish government.
- Optimize your compliance strategy
- Reduce long-term costs
- Protect your market position
Turkey REACH Registration Service Delivery Process
Our service delivery timeline is given below. To know more about how to get started email us at Turkey-REACH@qpregulatory.com or fill up contact form so we can get in touch with you as per your requirements
Step | Timeline | Details |
Consultation | Day 1 | Share CAS numbers & tonnage |
Audit | Days 1–2 | Check SIEF & LR status |
Quote | Day 3 | Fixed pricing |
Pre-registration | Days 3–5 | Secure access |
SIEF Management | Ongoing | Cost optimization |
Temporary Registration | By September 2026 | Maintain access |
Full Registration | By December 2032 | Maintain access |
Frequently Asked Questions (FAQs)
Q1: I missed the original pre-registration in December 2020. Can I still export to Turkey?
Yes, but action is urgent.
You can still submit a Late Pre-registration until October 31, 2025. Once submitted, your substance can legally remain on the Turkish market while you proceed with full registration.
After this deadline, new pre-registrations will not be accepted, and your products will be blocked at Turkish customs.
Q2: What does an Only Representative (OR) do under KKDIK?
An Only Representative (OR) acts as your legal entity in Turkey, taking over all regulatory responsibilities from the importer.
Key responsibilities include:
- Substance registration in the KKS system
- Direct communication with Turkish authorities
- Managing all compliance obligations
- Protecting your confidential business information
This allows your Turkish customers to operate as downstream users, significantly reducing their regulatory burden.
Q3: What is the difference between an OR and a Lead Registrant (LR)?
|
Role |
Function |
|
Représentant unique (RO) |
Represents non-Turkish companies legally in Turkey. One OR per company. |
|
Lead Registrant (LR) |
Leads the joint registration dossier for a specific substance. One LR per substance. |
GPC Regulatory can perform both roles seamlessly for your business.
Q4: How much does KKDIK registration cost?
Costs depend on several factors:
- Annual tonnage band (1–10, 10–100, 100–1000, 1000+ t/y)
- Availability of existing toxicological data
- Whether a Lead Registrant already exists for your substance
- Data-sharing fees within the SIEF (MBDF)
Cost-saving tip: Using existing EU REACH data can significantly reduce your expenses.
Contact GPC Regulatory for a custom quote within 3 business days.
Q5: Can I use my EU REACH registration for KKDIK compliance?
No, KKDIK is a separate legal framework.
However:
- Data from your EU REACH registration can often be reused
- Duplicate testing can be avoided
- Your overall compliance costs can be significantly reduced
GPC Regulatory specializes in data transfer and adaptation from EU REACH to KKDIK.
Q6: What is Temporary Registration and why is it important?
Temporary Registration is a critical compliance mechanism introduced by the Turkish Ministry.
It allows you to:
- Submit limited data (Annex 1 – physicochemical properties only)
- Maintain continued market access after the 2026 deadlines
- Buy time to generate full toxicology data for final registration
Without Temporary Registration, most companies will lose market access while preparing full dossiers.
Q7: What happens if there is no Lead Registrant for my substance?
A Lead Registrant (LR) must be appointed for joint submission.
If no LR exists:
- Your registration will be delayed
- Costs may increase significantly
- You cannot complete the registration process
GPC Regulatory can act as your Lead Registrant, ensuring:
- Faster submission to meet deadlines
- Cost control through fair data sharing
- Efficient coordination with all SIEF members
Q8: What are the penalties for non-compliance with KKDIK?
Non-compliance can lead to:
- Immediate loss of market access
- Customs rejection of all shipments
- Contract cancellations by Turkish buyers
- Significant financial losses
- Reputational damage in the Turkish market
Compliance is not optional—it is essential for business continuity in Turkey.
Q9: How long does the KKDIK registration process take?
|
Service |
Typical Timeline |
|
Late Pre-registration |
48 hours |
|
Temporary Registration |
Before July/September 2026 deadlines |
|
Full Registration |
1–3 years (depending on data availability) |
Starting early allows you to secure better data-sharing costs and avoid last-minute delays.
Q10: Why should I choose GPC Regulatory as my compliance partner?
GPC Regulatory delivers:
- Unmatched experience: 1,200+ dossiers managed, 400+ Lead Registrant roles
- Speed: 48-hour Late Pre-registration filing
- Cost savings: Existing data holdings reduce your expenses
- Local presence: Physical office in Turkey with direct MoEUCC access
- Certified experts: KDU-certified SDS authors and assessors on staff
- End-to-end service: From pre-registration through final registration in 2030
We handle the regulation—you focus on your business.
Voulez-vous en savoir plus sur la réglementation turque en matière de produits chimiques ?
Get in Touch with Our Regulatory Expert
Turquie REACH
Consultez notre document sur la réglementation des produits chimiques en Turquie !




